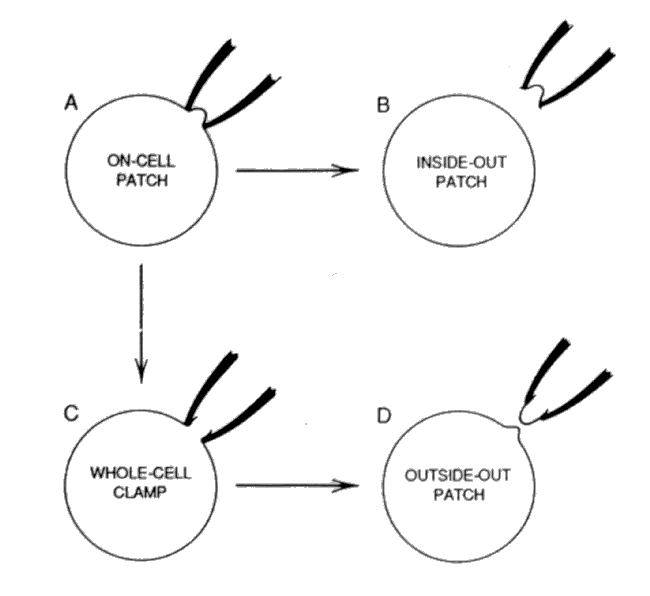
USE OF SIMPLE ELECTRICAL EQUIVALENT CIRCUITS.*. The whole-cell (WC) configuration is obtained by rupturing the CAP.

Establishment of the Dual Whole Cell Recording Patch Clamp Configuration for the Measurement of Gap Junction Conductance. The development of the patch clamp technique has enabled investigators to directly measure gap junction conductance between isolated pairs of small cells with resolution to the single channel level. The dual patch clamp recording technique requires specialized equipment and the acquired skill to reliably establish gigaohm seals and the whole cell recording configuration with high efficiency. This chapter describes the equipment needed and methods required to achieve accurate measurement of macroscopic and single gap junction channel conductances. Inherent limitations with the dual whole cell recording technique and methods to correct for series access resistance errors are defined as well as basic procedures to determine the essential electrical parameters necessary to evaluate the accuracy of gap junction conductance measurements using this approach. Key words. Gap junction conductance.

The whole-cell voltage-clamp configuration was obtained using a KCl-based pipette solution (in mM). Conventional tight seal whole-cell patch-clamp in voltage-clamp mode was performed using an Axopatch 200A amplifier with a.
Transjunctional voltage. Patch clamp. Dual whole cell configuration. Series resistance. Membrane potential. Channel conductance. Perforated patch.
Whole Cell Patch Clamp for Investigating the Mechanisms of Infrared Neural Stimulation. Infrared nerve stimulation has been proposed as an alternative to electrical stimulation in a range of nerve types, including those associated with the auditory system. This protocol describes a patch clamp method for studying the mechanism of infrared nerve stimulation in a culture of primary auditory neurons.
- Curr Protoc Pharmacol. 2012 Jun;Chapter 10:Unit 10.15. Whole-cell configuration of the patch-clamp technique in the hERG channel assay to predict the ability of a compound to.
- Patch clamp recordings in the whole cell configuration, and single channel recordings in the cell attached configuration. With conventional patch clamp this is difficult because of the tiny dimensions of the pipette tip.

Date Published: 7/3. Issue 7. 7; doi: 1.
Keywords: Neuroscience, Issue 7. Biomedical Engineering, Neurobiology, Molecular Biology, Cellular Biology, Physiology, Primary Cell Culture, Biophysics, Electrophysiology, fiber optics, infrared neural stimulation, patch clamp, in vitro models, spiral ganglion neurons, neurons, patch clamp recordings, cell culture. Brown, W. G., Needham, K., Nayagam, B.
This website uses cookies to deliver its services as described in our Cookie Policy. By using this website, you agree to the use of cookies. Patch Pipettes The patch clamp technique is used in electrophysiological research to study the electrical activity. The whole cell patch configuration is achieved when additional negative. Whole-Cell Voltage Clamp of Isolated Heart Cells. This article briefly summarizes the use of this variant of the patch-clamp technique in isolated. The whole-cell configuration is subsequently attained by. Whole-cell patch and perforated patch allow the researcher to study the electrical behavior of the entire cell. After the whole-cell configuration is formed. To achieve a loose patch clamp on a cell membrane.
Whole Cell Patch Clamp for Investigating the Mechanisms of Infrared Neural Stimulation. Infrared neural stimulation has been reported in a variety of peripheral and sensory neural tissue in vivo, with particular interest shown in stimulation of neurons in the auditory nerve. However, while INS has been shown to work in these settings, the mechanism (or mechanisms) by which infrared light causes neural excitation is currently not well understood. The protocol presented here describes a whole cell patch clamp method designed to facilitate the investigation of infrared neural stimulation in cultured primary auditory neurons. By thoroughly characterizing the response of these cells to infrared laser illumination in vitro under controlled conditions, it may be possible to gain an improved understanding of the fundamental physical and biochemical processes underlying infrared neural stimulation. The fields of neurophysiology and medical bionics rely heavily on techniques that allow controllable stimulation of electrical responses in neural tissue.
While electrical stimulation remains the gold standard in neural excitation, it suffers from a number of drawbacks such as the presence of stimulation artifacts when recording neural responses, and a lack of stimulation specificity due to the spread of current into surrounding tissue 1. The last two decades have seen the development of optically mediated stimulation techniques 2. Several of these techniques require modification of the target tissue, either via the addition of a particular molecule (e. Of particular interest therefore is infrared neural stimulation (INS), whereby neural tissue is excited by pulsed infrared laser light.
INS has the potential to overcome many of the shortcomings of electrical stimulation by enabling highly specific, non- contact stimulation of neural tissue 2. However, while INS has been successfully demonstrated in a variety of settings in vivo, the precise mechanism of excitation remains uncertain.
Some recent publications have shown progress towards uncovering the mechanism behind INS 5- 7. Rapid heating due to absorption of the laser light by water appears to play a key role. However, beyond this a consensus is yet to be reached. In addition, Albert et al. At this stage it is unclear how these mechanisms combine, or indeed whether there are further factors that are yet to be identified.
Although a small number of publications (references 5,7- 9) have investigated INS in vitro, the vast majority of work published in this field has been carried out in vivo (e. Infrared stimulation of auditory neurons has been an area of particular interest, owing to the potential applications in cochlear implants 1. While in vivo experiments are important to verify the effectiveness of the technique in various settings, the increased level of control afforded by in vitro studies is expected to lead to a more detailed understanding of the mechanism responsible for INS. This report describes the preparation of cultured spiral ganglion neurons for patch clamp investigations, as these can be used to study fundamental mechanisms while also linking to the large body of existing data from the auditory system. The patch clamp technique is an excellent tool for investigations of electrophysiological phenomena, providing a means of recording electrical activity in single cells and studying the contribution of the individual underlying currents. When this technique is applied to a stable in vitro preparation of primary neurons, such as cultured spiral ganglion neurons, it offers the opportunity to study in depth the mechanisms by which neural activity is controlled and manipulated.
The protocols specified in this work outline methods for investigating the effect of laser stimulation on the electrical properties of spiral ganglion neurons through patch clamp recordings. The approach is based on a fiber- coupled laser rather than a free- space laser, allowing safer operation as well as easier and more repeatable alignment without the need to modify the standard microscope configuration. On the basis of these protocols, it should be possible to conduct a wide range of experiments in order to more clearly determine the mechanism or mechanisms behind INS. Subscription Required. Please recommend Jo. VE to your librarian. Culture of Spiral Ganglion Neurons.
Sterilize small round (e. Transfer the sterilized coverslips into individual wells of a sterile 4- ring 3. Ensure that the coverslips do not float away from the bottom of the well. Prepare 5. 0 ml sterile Neurobasal media (NBM) for each neural culture: 4. A, 0. 5 ml N2 supplement, 1 ml B2. L- glutamine, and 0.
Note: Supplements can be frozen, stored at - 2. Refer to Whitlon et al.
Parker et al. 2. 4 for a demonstration of modiolus isolation. Aspirate any remaining poly- L- ornithine/laminin solution from the coverslips and wash briefly with NBM. Add 1. 50- 2. 00 . Note: up to 2. 0 coverslips can be prepared from an average litter of 8 rat pups. Four hours after plating neurons, aspirate the solution to remove cell debris and replace with 1.
Note: media may require daily replenishment to avoid dehydration. Return coverslips to the incubator until required for electrophysiological recordings.
Note: dissociated spiral ganglion neuron cultures can be used for electrophysiological experiments four hours after dissociation and for up to two days thereafter. Time in vitro should be taken into account during analysis of results. Replenish NBM every 2. Preparation for Patch Clamp Recordings. Prepare solutions.
Intracellular (micropipette) solution: 1. M K- gluconate, 7 m. M KCl, 1. 0 m. M HEPES, 0.
M EGTA, 2 m. M Na. ATP, 2 m. M Mg. ATP, 0. M Na. 2GTP (adjust to p. H 7. 3 with KOH; adjust to 2. Osmol/kg with sucrose).
Pass the solution through a sterile filter (0. This solution is made on the day of recording. Prepare recording micropipettes with a resistance of 2- 6 M. We use a CO2 laser puller (P- 2. Sutter Instruments) and borosilicate glass (1. Prepare the laser.
This protocol is intended for use with a fiber- coupled laser, such as the 1,8. Infrared Nerve Stimulator from Opto. Tech P/L. The optical fiber used for light delivery in our experiments is a 2. The patch cords were cut in half to produce two fiber pigtails (i. The effects of fiber core diameter and numerical aperture on laser induced temperature changes have been discussed in detail by Thompson et al. Cleave the tip of the light delivery fiber using standard techniques and ensure that the resulting tip is of high quality by observation with an optical microscope (i. Connect the light delivery fiber to the fiber- coupled output of the stimulation laser using an appropriate through connector (e.
Thorlabs ADAFC2). Measure the output laser power from the cleaved tip of the light delivery fiber using an appropriate instrument (e. Coherent Field. Mate with LM- 3 detector head).
It is recommended to check the laser power every time the light delivery fiber is cleaved or a significant adjustment is made to the laser (e. It is important to be able to accurately determine the angle . This angle can be measured by taking a photograph of the experimental arrangement and using image processing software (e. Image. J) to obtain the angle.
Digidata 1. 44. 0A, Molecular Devices) as shown in Figure 2. The digital output from the patch clamp data acquisition system should be connected to the laser via an external function generator, making it possible to specify laser pulse parameters independent of the data acquisition system. Alternatively this output can be connected directly to the laser driver (requiring the pulse length and repetition rate to be set by the data acquisition software). In either case, the signal used to trigger the laser should be connected back to an input of the data acquisition system to ensure that the timing and length of the laser pulses can be recorded concurrently with the electrophysiological signal. Patch Clamp Recordings for Investigation of INSFill the appropriate container of the perfusion system with extracellular solution and adjust the flow rate to provide perfusion of the bath at a rate of 1- 2 ml/min.
We use a gravity- fed system (aspirator bottle, pinch valve, and PE tubing) with an in- line heater to enable rapid heating of the solution, and a peristaltic pump to remove spent solution by suction. Place a coverslip with cultured cells into the recording chamber (bath) of an upright microscope. Using a high magnification water- immersion objective (e. X) and phase- contrast (e. A typical spiral ganglion neuron is phase- bright, round and approximately 1.
X) and locate the target neuron within the visual field. Move the light delivery optical fiber into position using the following procedure (or equivalent). Use the micropositioner to move the output fiber until the tip is close to the target neuron in both the horizontal and vertical planes.
The vertical position of the fiber tip can be verified by moving the objective up and down (scanning the focus). Switch back to the high magnification objective and position the tip of the optical fiber in its intended position next to the neuron (see Figure 2 inset).
 RSS Feed
RSS Feed
